History of the Atom (Evolution)
Atomic theory is the scientific theory of the nature of matter. The theory states that matter is made up of small particles called atoms. Prior to this theory, matter was thought to be able to be divided into any small quantity. The word atom is derived from the Greek atmos, meaning indivisible
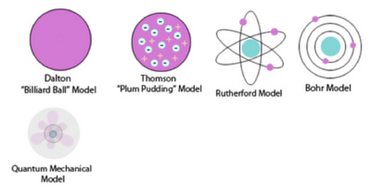
John Dalton (Dalton Model), in 1803, proposed that all matter is composed of atoms. This was not a completely new concept as the ancient Greeks (notably Democritus) had proposed that all matter is composed of small, indivisible (cannot be divided) objects. Dalton proposed a model where electrons and the nucleus were unknown.
After the electron was discovered by J.J. Thomson in 1897, people realised that atoms were made up of even smaller particles than they had previously thought. However, the atomic nucleus had not been discovered yet and so the “plum pudding model” was put forward in 1904. In this model, the atom is made up of negative electrons that sit with the positive charge particles, much like plums in a pudding. However, even with the Plum Pudding Model, there was still no understanding of how these electrons in the atom were arranged.
Rutherford carried out some experiments which led to a change in ideas around the atom. His new model described the atom as a tiny, dense, positively charged core called a nucleus surrounded by lighter, negatively charged electrons.There were, however, some problems with Rutherford's model: for example it could not explain the observation that atoms only emit light at certain wavelengths or frequencies.
Niels Bohr solved this problem by proposing that the electrons could only orbit the nucleus in certain special orbits at different energy levels around the nucleus.
Rutherford predicted (in 1920) that another kind of particle must be present in the nucleus along with the proton. He predicted this because if there were only positively charged protons in the nucleus, then it should break into bits because of all the positive charged protons repulsing off of each other. To make sure that the atom stays electrically neutral, this particle would have to be neutral itself. In 1932 James Chadwick discovered the neutron and measured its mass.
After the electron was discovered by J.J. Thomson in 1897, people realised that atoms were made up of even smaller particles than they had previously thought. However, the atomic nucleus had not been discovered yet and so the “plum pudding model” was put forward in 1904. In this model, the atom is made up of negative electrons that sit with the positive charge particles, much like plums in a pudding. However, even with the Plum Pudding Model, there was still no understanding of how these electrons in the atom were arranged.
Rutherford carried out some experiments which led to a change in ideas around the atom. His new model described the atom as a tiny, dense, positively charged core called a nucleus surrounded by lighter, negatively charged electrons.There were, however, some problems with Rutherford's model: for example it could not explain the observation that atoms only emit light at certain wavelengths or frequencies.
Niels Bohr solved this problem by proposing that the electrons could only orbit the nucleus in certain special orbits at different energy levels around the nucleus.
Rutherford predicted (in 1920) that another kind of particle must be present in the nucleus along with the proton. He predicted this because if there were only positively charged protons in the nucleus, then it should break into bits because of all the positive charged protons repulsing off of each other. To make sure that the atom stays electrically neutral, this particle would have to be neutral itself. In 1932 James Chadwick discovered the neutron and measured its mass.
