Hydrogen Ion:

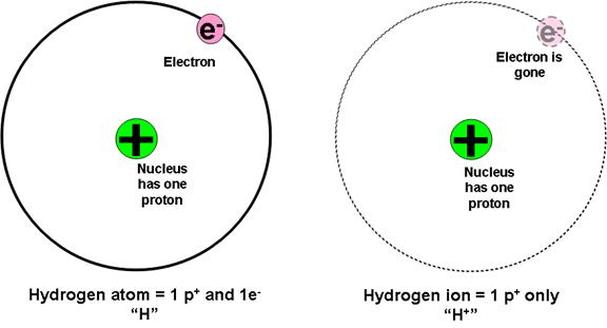
The atom of Hydrogen has one proton (positive charge) and one electron(negative charge) this is represented as 1p+ = 1e-
To make the Hydrogen atom "happy" with full shells, the electron is "donated" (lost) to create a +1 ion.
Types of Chemical bonding:
Ionic bonding is a type of chemical bond that involves the electrostatic attraction between oppositely charged ions. These ions represent atoms that have lost one or more electrons (known as cations) and atoms that have gained one or more electrons (known as an anion).
Metallic bonding is the strong attraction between closely packed positive metal ions and a 'sea' of delocalised electrons. The attraction between the metal ions and the delocalised electrons must be overcome to melt or to boil a metal.
A covalent bond is a chemical bond that involves the sharing of electron pairs between atoms. The stable balance of attractive and repulsive forces between atoms when they share electrons is known as covalent bonding.
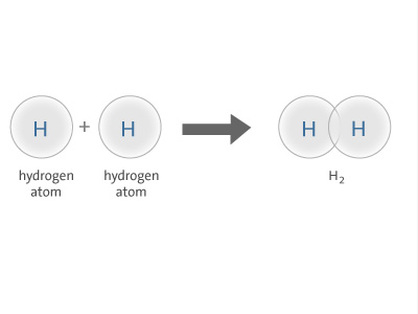
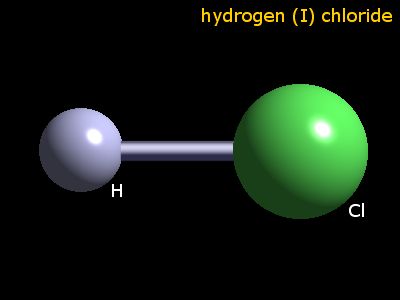
Hydrogen Covalent Bond : H2 Hydrogen Ionic Bond: HCl (Hydrochloric acid)
Hydrogen - H2 - Hydrogen is widely used for the
hydrogenation of vegetable and animal oils and fats. Hydrochloric acid is an important and widely used chemical. The largest end uses for hydrochloric acid are steel pickling, oil well acidizing, food manufacturing, producing calcium chloride, and ore processing.
To make the Hydrogen atom "happy" with full shells, the electron is "donated" (lost) to create a +1 ion.
Types of Chemical bonding:
Ionic bonding is a type of chemical bond that involves the electrostatic attraction between oppositely charged ions. These ions represent atoms that have lost one or more electrons (known as cations) and atoms that have gained one or more electrons (known as an anion).
Metallic bonding is the strong attraction between closely packed positive metal ions and a 'sea' of delocalised electrons. The attraction between the metal ions and the delocalised electrons must be overcome to melt or to boil a metal.
A covalent bond is a chemical bond that involves the sharing of electron pairs between atoms. The stable balance of attractive and repulsive forces between atoms when they share electrons is known as covalent bonding.
Hydrogen Covalent Bond : H2 Hydrogen Ionic Bond: HCl (Hydrochloric acid)
Hydrogen - H2 - Hydrogen is widely used for the
hydrogenation of vegetable and animal oils and fats. Hydrochloric acid is an important and widely used chemical. The largest end uses for hydrochloric acid are steel pickling, oil well acidizing, food manufacturing, producing calcium chloride, and ore processing.